Prioritize...
After you've finished this section, you should be able to:
- Define fossil fuels and give three examples of them, including how they were formed.
- Define combustion and qualitatively describe why burning these fuels leads to CO₂.
- Use your knowledge of the carbon cycle and its speeds, along with fossil fuels, to explain why burning these fuels disrupts the natural timescales of carbon storage and release in the Earth.
Read…
So, why is carbon such a big deal?
Well, carbon is essential in living organisms because it forms stable bonds with other atoms, creating many complex molecules. Insert chemistry flashbacks here! These complex molecules are the building blocks of life on Earth, forming everything from proteins and fats to carbohydrates and nucleic acids. A great example is DNA, the molecule that carries genetic information, which has a backbone made of carbon atoms. When these carbon-based molecules break apart and form bonds with oxygen to create carbon dioxide, they release energy that is critical for all life forms - from the tiniest microorganisms to the largest mammals.
In the carbon cycle, plants and tiny ocean organisms called phytoplankton play a crucial role by absorbing carbon dioxide from the air. Through the process of photosynthesis, a process that requires energy from the sun, they convert carbon dioxide and water into sugars (storage containers of energy initially from the sun) and release oxygen as a byproduct. You've probably heard how trees “clean” the air by taking in carbon dioxide and releasing oxygen. This process can be summarized by the chemical equation:
Plants return carbon to the atmosphere in four main ways: through cellular respiration (which breaks down sugars using oxygen, releasing energy and CO₂), when animals consume them, when they decompose after dying, and when they burn. In each of these processes, the sugars in plants are broken down, releasing energy, water, and carbon dioxide back into the air:
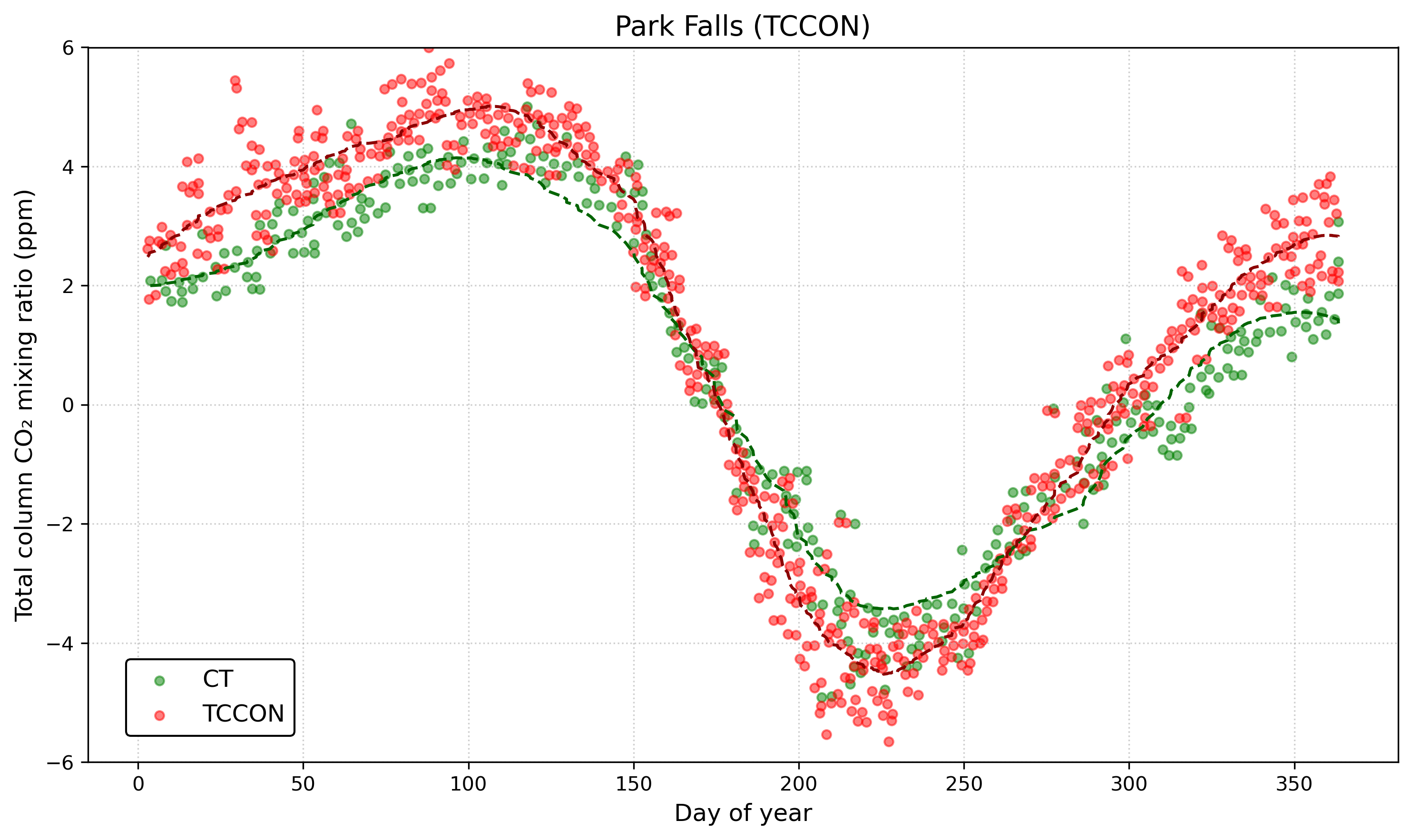
This release of carbon dioxide is part of the fast carbon cycle and is closely linked to how plants grow and change with the seasons. For example, in the Northern Hemisphere, atmospheric carbon dioxide levels go up in the winter when fewer plants are growing (and taking up CO₂) and more are decaying (releasing CO₂), and then they drop in the spring when plant growth picks up again. This yearly cycle shows how the Earth itself seems to “breathe” with the seasons. See the graph below, which shows CO₂ measurements in the air at Park Falls, Wisconsin. The time series is plotted as an anomaly (think back to lesson 1) relative to the annual mean. It is highest at the very left and very right of the graph, representing the colder, darker winter months in the Northern Hemisphere (“Day of Year” equal to 1 is January 1st), when very little photosynthesis occurs. It then rapidly drops starting around day 110-120, which corresponds to mid-to-late April as trees leaf out and ground vegetation grows robustly. It bottoms out around day 225 or so, which corresponds to mid-August, the very end of the growing season, when plants in the area start to become dormant.
So, under natural conditions, the interplay between the fast and slow carbon cycles helps maintain a balanced level of carbon in the atmosphere, land, plants, and ocean. However, any disruption in one area of this balance can create ripple effects across the others!
This is where scientists grow concerned. Currently, human activities are significantly altering the carbon cycle in ways that amplify these imbalances. While we’ll dive into topics like deforestation (i.e., removing plants as natural carbon vacuums) later, let’s first focus on how fossil fuels fit into the picture.
Fossil fuels—like coal, oil, and natural gas—are energy sources that trace their origins back to ancient organic matter, formed millions of years ago. They’re called “fossil” fuels because they come from the preserved remains of long-dead plants and animals, stored deep within the Earth’s layers. It’s like nature’s slow cooker, but instead of a delicious stew, we get coal, oil, and gas when the dial is set to “geologic time.” I think I’ll stick to using my crockpot for chili, though!
Coal, for instance, formed from the dense, swampy forests that existed during the Carboniferous period, around 300 million years ago. The name Carboniferous literally means “coal-bearing,” from the Latin carbō (“coal”) and ferō (“carry”), and refers to the many coal beds formed globally during that time! As these plants died, they became buried in waterlogged environments where oxygen was scarce, slowing their decay. Over time, layers of sediment piled on top, fully cutting the dead plants off from the atmosphere. Eventually, increasing pressure and heat gradually transformed this plant material into coal.
Oil and natural gas, on the other hand, originated from tiny marine organisms—microscopic plants and animals—that drifted to the ocean floor when they died. As layers of mud and sediment buried them, similar forces of heat and pressure worked on this organic matter. However, unlike coal, the combination of factors caused the formation of liquid oil and gaseous natural gas. Over millions of years, these hydrocarbons migrated through porous rocks, eventually getting trapped in pockets deep beneath the Earth's surface.
So why does it matter when we extract these long-buried materials? Fossil fuels are packed with energy because they’re made of hydrocarbons—molecules composed of carbon and hydrogen atoms. The name says it all: hydrocarbons. When we burn these fuels in the presence of oxygen, the bonds between the carbon and hydrogen atoms break, and then the carbon forms strong bonds with oxygen, forming carbon dioxide and releasing energy that had been stored for millions of years in the weaker carbon and hydrogen bonds. The burning process, called combustion, transforms hydrocarbons into carbon dioxide (CO₂) and water vapor (H₂O), while releasing energy as heat—and sometimes light, like the flame on a gas stove or the thrust from a rocket engine.
For instance, when methane (CH₄), the main component of natural gas, combusts, it follows this chemical equation:
Combustion & Incomplete Combustion | Environmental Chemistry | FuseSchool
- Introduction
The video opens with the FuseSchool logo and the title “Combustion & Incomplete Combustion.”
An animated flame appears, symbolizing the topic of combustion. - Complete Combustion
What Combustion is:
The difference between complete and incomplete combustion.
The products of the two types of combustion.
The effects of the products of combustion.
Exothermic Reaction
Gives out heat
Fuel stores potential energy.
Examples of fuels, natural gas, methane.
Methane undergoes complete combustion
An animation shows a hydrocarbon fuel (like methane) reacting with oxygen.
Equations: CH₄ + 2O₂ → CO₂ + 2H₂O
Visuals depict the release of carbon dioxide (CO₂) and water (H₂O), with bright flames indicating the complete combustion process.
The text explains that complete combustion occurs with sufficient oxygen, producing CO₂ and H₂O. - Incomplete Combustion
Methane undergoes incomplete combustion.
The animation changes to show limited oxygen availability.
Equations: 2CH₄ + 3O₂ → 2CO + 4H₂O and CH₄ + O₂ → C + 2H₂O
Visuals depict the release of carbon monoxide (CO), water, and carbon (soot), with dull flames and smoke indicating incomplete combustion.
Water vapor is harmless.
The text explains that incomplete combustion occurs with insufficient oxygen, producing CO and soot. - Environmental and Health Impacts
Animations show the harmful effects of carbon monoxide and soot on health and the environment.
Visuals depict carbon monoxide binding to hemoglobin in the blood, reducing oxygen transport, leading to health issues.
Carbon monoxide is colorless, odorless.
Faulty gas appliances
It is important
Gas fires and boilers regularly serviced.
Unburnt carbon is a particulate.
A pollutant in the air.
Soot particles are shown to cause respiratory problems and contribute to air pollution. - Summary
A recap of the key points: Complete combustion needs enough oxygen, producing CO₂ and H₂O. Incomplete combustion lacks oxygen, producing CO and soot.
Combustion is burning, usually in air or oxygen.
Complete combustion of hydrocarbons produces carbon dioxide and water.
Incomplete combustion produces either carbon monoxide and water, or carbon and water.
The products of combustion have various effects.
Left to the Earth’s natural devices, this carbon from fossil fuels would slowly enter the atmosphere over millions of years due to volcanic activity or rock weathering (hey, it’s that slow cycle!). However, when humans extract and burn fossil fuels, things speed up dramatically—essentially moving carbon from the slow cycle into the fast cycle. This rapid release of carbon into the atmosphere is what concerns scientists most about climate change. It disrupts the natural balance of the carbon cycle, causing a spike in atmospheric carbon dioxide -- a potent greenhouse gas -- over a relatively short span of geological time (instead of talking thousands, millions, or billions of years, we're now talking tens of years!).