Prioritize...
At the completion of this section, you should be able to:
- Describe how ocean warming and high heat capacity impact the rate of temperature increase in oceans compared to the atmosphere.
- Explain the process of ocean acidification, its effects on marine organisms that rely on calcium carbonate, and the potential economic implications for human food security.
Read...
In the last section we found that, since the late 1800s, global average surface air temperatures have increased about 1 degree Celsius (about 2 degrees Fahrenheit). You might wonder, "Why worry about a small bit of warming?" That's a fair question! This recent rise is significant because, since the last ice age ended about 10,000 years ago, global average surface air temperatures have fluctuated by only about 1.7 degrees Celsius (close to 3 degrees Fahrenheit) in total. In contrast, today’s warming—over a degree Celsius in just about a century, most of it since 1980—marks an unusually fast change.
What makes this rapid shift even more noteworthy is our global context: when Earth was last this warm, it wasn’t home to over 7 billion people, nor had human-built infrastructure that spans almost every continent. Our ancestors shaped their communities, agricultural practices, and economies around a relatively stable climate over the past several centuries. But as our planet continues to warm, those climate norms—the foundation of modern civilization—are shifting. And while a couple of degrees may seem small, let’s consider a familiar analogy: human body temperature. Our normal body temperature is 98.6°F, but a rise of just 2 or 3 degrees signals a fever, leaving us feeling unwell. So, even small changes in temperature can have big impacts!
Ocean Warming and Acidification
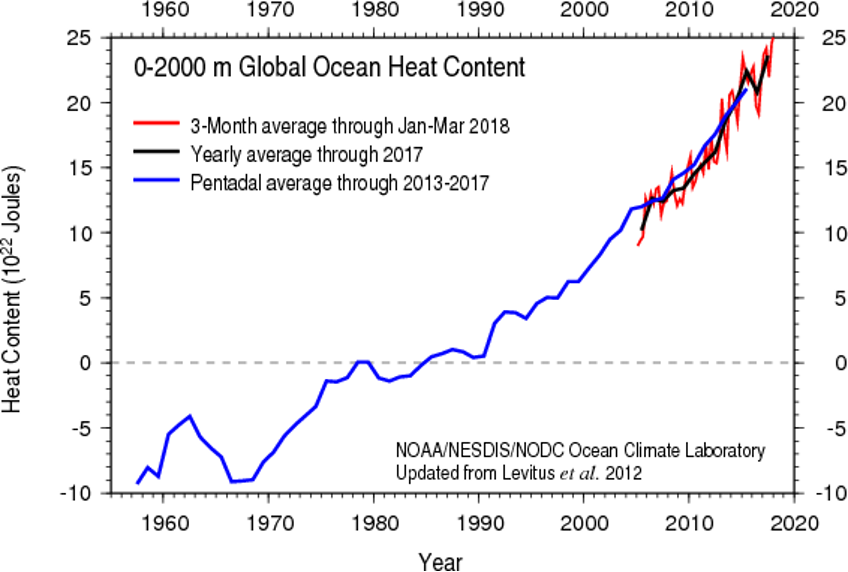
For starters, the atmosphere isn't the only part of the Earth system that's warming. The hydrosphere, which includes all of Earth's water—oceans, rivers, lakes, ice, and even water vapor in the air—is also changing as the climate warms. The graph below shows global ocean "heat content" from the ocean surface down to a depth of 2000 meters since the late 1950s. “Ocean heat content” refers to the total amount of heat stored within this layer, so it not only includes the surface (or skin) temperature of the ocean, but the temperature all the way down to 2 km – or about 5 Empire State buildings!
As oceans absorb more heat due to an intensified greenhouse effect, they’re warming, but at a slower rate than the atmosphere. This difference is due to the high heat capacity of water—a property that requires more energy to raise the temperature of water than air. Think of it this way: on a hot summer day, the air in your backyard heats up fast, while your (non-heated) pool stays cool by comparison. This is because water absorbs and retains heat more efficiently than air, allowing the oceans to take in large amounts of energy without a big jump in temperature. So, while both the atmosphere and oceans are warming, the ocean’s ability to hold heat acts as a buffer, slowing the rate of temperature rise in the water.
Not only are the oceans warming, they're also becoming more acidic. This increase in acidity refers to the rise in the concentration of hydrogen ions in the ocean, which lowers its pH level. Oceans play a pivotal role in Earth's carbon cycle, acting as a significant carbon sink that helps regulate the global climate. In fact, if you remember from our carbon cycle discussion, they are the largest reservoir of carbon on the planet, storing a whopping 50 times more carbon than the atmosphere! The process through which oceans absorb carbon dioxide (CO2) from the atmosphere is complex and multifaceted. When CO2 from the atmosphere dissolves in seawater, it reacts with water to form carbonic acid, which then breaks down into bicarbonate and hydrogen ions. This series of reactions helps to reduce the concentration of CO2 in the atmosphere, thereby mitigating the greenhouse effect and global warming to some extent. However, this increased uptake of CO2 by the oceans is not without consequences.
When carbon dioxide dissolves in ocean water, it forms carbonic acid, which gradually leads to ocean acidification (a decrease in pH). The graph below shows global ocean pH changes from 1982 to 2021, steadily drifting towards lower values. Now, you might think, “We only went from a pH of 8.125 to 8.055—a shift of just 0.07 units—so what’s the big deal?” But here’s the key: pH is measured on a logarithmic scale, meaning that small shifts translate to big changes in acidity. In this case, a drop of 0.07 pH units represents an 18% increase in ocean acidity!
Explore Further...
Head here to learn a bit more about pH scales to refresh your memory from high school chemistry!

So, what does this drop in ocean pH actually mean? On one hand, photosynthetic algae—key oxygen producers and essential players at the base of the marine food web—might initially benefit. These algae rely on carbon dioxide for photosynthesis (the process of converting sunlight and CO₂ into energy), so as more CO₂ dissolves in the ocean, they could gain more resources to grow.
However, the story is different for many other marine organisms. Lower pH means a change in the ocean’s chemistry, particularly making it harder for species that use calcium carbonate to build their shells and skeletons. This includes corals, mollusks like clams and oysters, and certain types of plankton, all of which are essential to the marine food chain. As the water becomes more acidic, these organisms struggle to grow and maintain their structural defenses, leaving them vulnerable to disease and other environmental pressures. The effects ripple throughout the ecosystem: weakened corals and shellfish impact larger fish and marine animals that depend on them for food. It can also have significant "downstream" impacts on fisherman livelihood and even our pocketbooks (see the diagram below).
Considering that approximately one billion people globally (about 1 in every 8 humans) depend on seafood as their primary source of protein, the health of these marine species is not just an environmental concern but also a crucial economic and food security issue.
