Putting the Pieces Together
Putting the Pieces TogetherPrioritize...
After reading this section you should be able to:
- Relate the concentrations of the "big four" greenhouse gases to one another.
- Explain in simple terms why CO₂ is the most important greenhouse gas, and why the properties of the other three gases make them important, even if their concentrations are much smaller.
Read...
We've just covered how human activities have driven up concentrations of methane, nitrous oxide, and halogenated gases (HGs). However, even with these increases, their concentrations are still much lower compared to carbon dioxide. Take a look at the table below, where the concentrations are listed in "parts per billion." It’s clear that CO₂ dominates by a wide margin. To put it in perspective, if fluorinated gases were a single person, they’d be like one person among four sold-out games at Beaver Stadium, while carbon dioxide would represent everyone else in the stands! This massive difference is a key reason why CO₂ often takes center stage in discussions about greenhouse gases and their impact on climate.
| Constituent | Concentration (ppb) |
|---|---|
| CO2 | 400,000 |
| CH4 | 1,900 |
| N2O | 320 |
| HGs | 1 |
It’s important to point out that while methane, nitrous oxide, and halogenated gases may be present in much smaller concentrations than carbon dioxide, the impact of these gases on emission and absorption of atmospheric infrared radiation for a fixed increase in concentration is far greater than for carbon dioxide. The physics behind this is a bit complex and might be better suited for a dedicated physics class, but in simple terms, it has to do with the molecular structure of these gases and the wavelengths at which they emit and absorb infrared radiation. Averaged all methane and carbon dioxide molecules in the atmosphere, it is as if one molecule of methane is 28 times more effective at absorbing and emitting infrared radiation than one molecule of carbon dioxide. So even though their concentrations are smaller—sometimes drastically so—we definitely can’t overlook their role as their concentrations change. In our atmosphere, a little bit of something can go a long way!
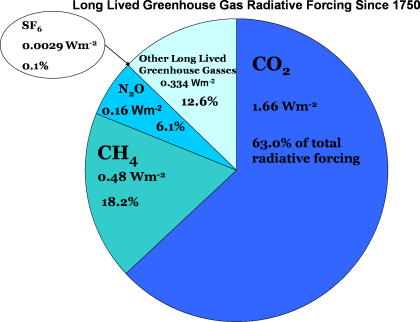
When you factor in both the absolute concentration of these gases and their relative ability to emit and absorb infrared radiation at different wavelengths, you get a clearer picture of their overall impact. The chart below shows how much additional radiation has been added to the climate system since 1750 due to these greenhouse gases, relative to a perfect balance of incoming and outgoing energy.
Roughly two-thirds of the warming caused by human activities can be attributed to carbon dioxide, while another 20% or so comes from methane. The rest is due to nitrous oxide and other greenhouse gases, including halogenated gases. So, while CO₂ might not be as potent on a molecule-for-molecule basis, its sheer volume in the atmosphere—thanks to fossil fuel combustion—still makes it the “top dog” among greenhouse gases.
The end result of these increases in greenhouse gases is that the atmosphere is now retaining about one percent more energy compared to pre-industrial times. Now, that might not sound like much, but when you remember that the atmosphere absorbs around 240 W/m² of energy from the sun, that extra one percent adds up! It’s a key factor in explaining why the planet has warmed nearly 1 degree Celsius (1.8 degrees Fahrenheit) globally since 1970. That small shift in energy balance has big consequences!