Lesson 3: Why Energy Drives Everything
Lesson 3: Why Energy Drives EverythingMotivate
Understanding energy is fundamental to understanding the climate system. In simple “physics” terms, energy can be described as the capacity to perform work. Practically, think about it this way. You and I need to move and eat and breathe – all of this requires us to do work and requires us to expend energy to do so. In fact, the concept of energy is so deeply intertwined with our physical universe that it becomes challenging to imagine a world without it. Everything we do, from driving a car to turning on a lightbulb, involves the conversion or transfer of energy in various forms. Therefore, energy is a concept that goes beyond this class (or any of your other science classes, for that matter!).

Overview of Energy
Overview of EnergyPrioritize...
By the end of this section, you will:
- understand the three fundamental principles of energy as they relate to the climate system
- recognize the different types of energy that play crucial roles in Earth's processes
- grasp how these energies are transferred and transformed according to the laws of thermodynamics
Read...
Overview
Energy is the ability to do work or cause change. It’s what makes things happen—whether it’s moving objects, heating up your food, lighting up a room, or even allowing you to run or think. Energy comes in many forms, like heat, light, electricity, and motion, and it can change from one form to another. For example, when you eat food, your body converts the energy stored in that food into the energy you need to move, grow, and stay warm. Without energy, nothing can move or change in the world around us.
When talking about energy to understand why the climate system behaves like it does, there are three fundamental principles we need to understand:
- Energy can be stored.
- Energy can move from one piece of matter to another.
- Energy can exist in different types and can transform between these types.
These three concepts also lay the groundwork for the First Law of Thermodynamics. According to this law, the total amount of energy within a closed system remains constant during all processes of energy movement and transformation.
Hmmm. That’s a bit of a mouthful – how can we frame this more simply? Let’s try this: Energy is never created nor destroyed, but it can move around and change forms. You may have also heard this called the “conservation of energy.” It’s worth noting the language “closed system” – this means a hypothetical box where energy cannot enter or exit. The grandest way to think about this is the universe – if we could place it in a giant box (a funny -- and very optional -- Futurama episode to watch), all energy within the box must remain constant because we assume no energy can enter or escape the universe.
Types of Energy
In the study of energy, we encounter a diverse array of “types” (sometimes referred to in textbooks as “forms”), each with unique characteristics and roles in the physical world. There are numerous forms of energy (ask a physicist for some of the more complicated ones!), but we will focus on four important ones here.
- Radiant Energy – the energy associated with electromagnetic radiation. As humans, we generally consider this manifesting as light – after all, it is the energy form that allows us to perceive the world around us (think visible light to our eyes, infrared heat from a fireplace, etc.). But as we will see very soon, it will also be critically important for getting energy into and out of (and moving it long distances around) the Earth system.
- Kinetic Energy - associated with motion, is evident in everything from the swaying of leaves in the wind to the bustling activity of a city. Any bit of matter, no matter how big or small or how light or how dense, possesses kinetic energy when it moves.
- Gravitational Potential Energy - determined by an object's height above the Earth's surface. It is called potential because it could be converted to kinetic energy if it starts falling toward the ground. Roller coaster aficionados know how potential energy and kinetic energy can be converted back and forth between one another – coasters tend to move slowly when very high and then move faster as they are closer to the ground.
- Internal energy - a composite of factors about a piece of matter itself (i.e., the atmosphere), such as its temperature, pressure, and chemical makeup.
What ties these diverse forms of energy together in the Earth system is their propensity for transfer and conversion. These transfers and conversions among various types of energy are the driving forces behind virtually every natural phenomenon. Therefore, we need to understand this to understand the climate system. While the First Law of Thermodynamics assures us that the total amount of energy in a system remains constant, it is important to recognize that not all energy is equally likely to undergo transfer or conversion. This leads us to the Second Law of Thermodynamics. According to this law, heat always flows spontaneously from hotter to colder regions of matter. A way to think about this that I think is somewhat intuitive is that heat always flows “downhill” if you think of a hill where the top represents warmth, and the valley represents cold.
What does this mean in practice? Let’s say I have a glass of ice cubes on my kitchen table. We know from personal experience that over time, those ice cubes will melt, and the resulting liquid will eventually become lukewarm. That is because the surrounding energy of the room – which is hopefully above freezing -- flows “downhill” to heat the ice cubes, melt them, and bring the water’s temperature in equilibrium with everything else around it. Left by itself, energy will always try to reduce its own order or organization itself, a concept known as “entropy.”

But wait a minute, maybe you should be skeptical of that. After all, if that’s the case, how do air conditioners and refrigerators function? After all, the whole reason they exist is to keep things cold, even when everything else around them is hot! Well, they can exist because they keep things cold (make heat flow “uphill” away from the inside of the freezer) by rearranging energy elsewhere (in their case, using electrical energy to push this heat up the hill). Similarly, we know that parts of the planet are cold and parts of the planet are warm. I know if -- for some reason -- I really, really, really want to feel an icy wind blow in my face, I can be essentially guaranteed that if I head to the South Pole. The second law of thermodynamics tells me that for this temperature imbalance between balmy Cancun, Mexico and frigid Antarctica to be maintained, energy must be being actively rearranged within the Earth system. While we will discuss all these forms of energy throughout this course, we start by focusing on radiant energy. Essentially, all the energy-sustaining life on our planet arrives in the form of radiation from our nearest star – the sun. All five components of the climate system consistently release radiation: 24 hours a day, 7 days a week. How this radiant energy is received, stored, moved, and transformed can teach us so much about the Earth’s climate! Read on.
Quiz Yourself…
Shedding Light on the Electromagnetic Spectrum
Shedding Light on the Electromagnetic SpectrumPrioritize…
At the completion of this section, you should be able to:
- define radiation, wavelength, and micron.
- discuss the organization of radiation in the electromagnetic spectrum by wavelength, including knowing which types of radiation have longer wavelengths and which have shorter wavelengths, for example).
Read…
So, we’ve now discussed that of the forms of energy, radiant energy (i.e., radiation) plays a central role. If you think of “radiation” and immediately conjure up images from science fiction movies or cautionary stories about nuclear energy, I can understand why! From a scientific perspective, radiation refers to the emission, transfer, and absorption of energy in the form of electromagnetic waves through space or a medium. It serves as a fundamental mechanism for transporting and exchanging energy within Earth's climate system. This encompasses solar radiation, which provides the energy that drives weather patterns and climate, as well as terrestrial radiation emitted by the Earth and its atmosphere.
Origins of Electromagnetic Spectrum of Radiation
During your science classes, you probably explored the electromagnetic spectrum of radiation. However, have you ever wondered about its origins? First, it's essential to recognize that all matter consists of atoms (the fundamental building blocks) and molecules (combinations of atoms). Within these already small structures exist even tinier particles with positive and negative charges, protons and electrons. Molecules tend to rotate andsibrate, and the electrons within them can move from lower energy orbitals to higher ones and vice versa. These movements of and within molecules lead to accelerations of the protons and electrons. Without delving into intricate details, physics teaches us that accelerated charges are the source of electromagnetic waves. It is amazing to think that all of the rotating water molecules around me (and most of the water molecules are indeed rotating!) are producing radiation, much of which strikes and is absorbed by my skin.
Another way to think about this is to imagine your hand as a vibrating molecule and a pond as the surrounding medium through which a wave propagates. Imagine moving your hand rapidly back and forth in the water – this generates waves that ripple out away from your hand and across the surface of the pond. Similarly, oscillating protons and electrons emit energy ripples known as electromagnetic "waves," and these waves propagate in every direction away from the accelerated charges whatever the surrounding medium might be, including the vacuum of space. These waves exhibit both electric and magnetic characteristics, hence the term “electromagnetic wave, or electromagnetic radiation, to describe them." The word “electromagnetic” is commonly abbreviated as “EM.”
Wavelengths
So, what gives rise to the different types of EM waves that form the complete spectrum? First, let's discuss the various categories of EM radiation based on wavelength. The wavelength of any wave is the distance from one identical point on the wave to the next, such as from crest to crest. Let's revisit our earlier analogy with the pond. When you move your hand through the water slowly, you generate a few waves with long wavelengths. Conversely, if you move your hand rapidly, you produce numerous waves with much shorter wavelengths. This same principle applies to an oscillating charge. When the oscillation is exceptionally fast (referred to as high frequency), the resulting EM radiation will have a short wavelength. Conversely, if the oscillation occurs at a slower rate (entailing a lower frequency), the electromagnetic waves will consist of longer wavelengths.
Now, the frequencies at which of charges in molecules can oscillate is primarily determined by the type and arrangement of atoms that compose the molecules.The temperature of the medium in which the molecules are situated subsequently determines how many molecules are oscillating at each frequency. As the temperature of the medium goes up, higher frequency oscillations within the molecules occur Why? The temperature of any piece of matter is determined by the kinetic energy of the atoms or molecules that compose the material. Remember, kinetic energy is a measure of the energy of a substance due to translation through space – we usually think about it in terms of throwing a ball or a moving train, but atoms and moleculesare tiny forms of matter and they too, can be moving through space! The higher the temperature, the faster these particles are moving. When faster moving atoms and molecules bang into each other, they are able to transfer more kinetic energy into the rotations, vibrations, and orbital excitations within the colliding particles, which correspond to higher frequency oscillations. Higher frequency oscillations, in turn, correspond to smaller wavelengths of the EM waves they generate. Conversely, as the temperature of a collection of atoms and moleculesdecreases, the frequencies induced by colliding particles decreases, and the wavelengths emanating from the collection of particles increase.
Explore Further…
Take a few minutes to explore how to measure and compare wavelengths by completing the Describing a Wave activity.
A Caveat
Extremely high-frequency EM radiation emissions (e.g., gamma rays) necessitate an additional mechanism to produce them, which goes beyond the scope of what's necessary for this course as these emissions are not important for understanding weather and climate.
Complete Spectrum of Electromagnetic Radiation
Now that we've addressed that caveat, let's examine the complete spectrum of electromagnetic radiation depicted below. Firstly, it's important to recognize the vast range of wavelengths that different types of electromagnetic radiation encompass — from hundreds of meters down to the dimensions of an atom's nucleus. Additionally, it's worth acknowledging that visible light falls within the category of electromagnetic radiation, albeit occupying just a minuscule portion of the entire spectrum. This fact underscores that our eyes are effectively blind to nearly all forms of electromagnetic radiation.
Video: What it Light? Maxwell and the Electromagnetic Spectrum (3:55)
Video: Science at NASA: An Introduction to the Electromagnetic Spectrum (5:19)
Starting from the far end of the spectrum characterized by its longest wavelengths, often referred to as the “long-wave” segment, we encounter radio waves and microwaves, boasting wavelengths ranging from hundreds of meters to just a few millimeters (one-thousandth of a meter, or 10^-3 meters). As we delve into shorter wavelengths, we find measurements often expressed in micrometers, more commonly termed microns (one-millionth of a meter, or 10^-6 meters). Within this range, when wavelengths decrease to the scale of tens of microns, comparable in size to a bacterium or a virus, we classify these emissions as infrared, visible, and ultraviolet light. In the final stretch of the spectrum, characterized by exceedingly short wavelengths comparable in size to individual molecules or atoms, we encounter X-rays and gamma rays.

A detailed visual representation of various types of electromagnetic radiation, their wavelengths, frequencies, and the temperature at which they are most intensely emitted. It includes a horizontal illustration of the electromagnetic spectrum, starting from radio waves on the left to gamma rays on the right.
Penetration of Earth's Atmosphere: A horizontal bar at the top illustrates whether each type of radiation penetrates Earth's atmosphere. It is divided into sections marked “Y” (Yes) and “N” (No), indicating if the radiation type can reach Earth.
Radiation Types and Wavelengths: A red waveform visually depicts the wavelength of each radiation type, starting with long wavelengths for radio waves and shortening progressively to gamma rays. Below, several categories of radiation types are listed with their respective wavelengths in meters: Radio (10³), Microwave (10⁻²), Infrared (10⁻⁵), Visible (0.5×10⁻⁶), Ultraviolet (10⁻⁸), X-ray (10⁻¹⁰), and Gamma ray (10⁻¹²).
Approximate Scale of Wavelengths: Various objects are depicted to represent the approximate scale of each radiation type's wavelength: buildings (radio), humans (microwave), butterflies (infrared), needle points (visible), protozoans (ultraviolet), molecules (X-ray), atoms (gamma ray), and atomic nuclei (gamma ray).
Frequency: A horizontal bar denotes the frequency range (Hz) corresponding to each type of radiation, starting from 10⁴ Hz (radio waves) to 10²⁰ Hz (gamma rays).
Temperature of Most Intense Emission: A colored horizontal scale at the bottom represents the temperature (in Kelvin and Celsius) at which each type of radiation is most intensely emitted. It ranges from 1K (-272°C) for radio waves to 10,000,000K (~10,000,000°C) for gamma rays.
In the context of this course, our primary focus will be on infrared, visible, and ultraviolet radiation, as these are relevant for climate scientists studying how energy is transferred into, within, and out of the Earth system. Additionally, we'll delve further into the infrared spectrum in this lesson, which extends “beyond red.” Notably, a significant portion of the infrared spectrum, spanning approximately 3 to 100 microns, is called “terrestrial” or “longwave” radiation — radiation originating from Earth. This is because at the temperatures commonly observed on our planet, including those within Earth's atmosphere, the molecules that compose the Earth system emit EM waves mostly with these wavelengths.
Now that you are familiar with the terminology used to describe the various segments of the electromagnetic spectrum, it's imperative to explore the characteristics governing the emission of radiation. These properties are organized into what we can think of as the “four laws of radiation.” Let's delve into these further.
Quiz Yourself…
Laws
LawsPrioritize…
By the end of this section, you will be able to
- describe the three primary laws of electromagnetic radiation—Planck's Law, Wien's Law, and the Stefan-Boltzmann Law
- understand how the laws relate to the emission, distribution, and intensity of energy from different objects, including the Earth and the Sun.
Read…
For electromagnetic radiation, there are three primary “laws” that describe the type and amount of energy being emitted by an object. In science, a law is used to describe a body of observations. At the time the law is established, no exceptions have been found that contradict it. The difference between a law and a theory is that a law simply describes something, while a theory tries to explain “why” something occurs. As you read through the laws below, think about observations you've made in everyday life that might support the existence of each law.
- Planck's Law
Planck's Law can be generalized this way: Every object emits radiation at all times and at all wavelengths. Surprising? We know that the sun emits visible light, infrared waves, and ultraviolet waves, but did you know that the sun also emits microwaves, radio waves, and X-rays? Of course, the sun is a big nuclear furnace, so it makes sense that it emits all sorts of electromagnetic radiation. Planck's Law means that you emit radiation at all wavelengths, and so does everything around you! But, depending upon the temperature of the object emitting, the object emits more radiation at some wavelengths than others.
Now before you dismiss this statement out-of-hand, let me say that you are not emitting X-rays in any measurable amount. Simply put, your temperature is way too low to excite the high frequency oscillations in the charges that compose you necessary to produce them. That said, perhaps once every millennium a tiny, unmeasurable amount of X-rays emanates from you because of some low probability fluctuation in the particles that compose you.
Another common misconception that Planck's Law dispels is that matter turns on and turns off the radiation that it emits. Consider what happens when you turn off a light bulb. Is it still emitting radiation? You might be tempted to say “No” because the light is off and you no longer see visible radiation emanating from it. However, the temperature of the light bulb is not zero, and Planck's Law tells us that while the light bulb may no longer be emitting radiation that we can see, it is still emitting at all wavelengths. It first emits mostly near infrared radiation when first turned off . As the light bulb cools down to room temperature, emission moves primarily from the near infrared to the terrestrial infrared. Another example that you hear occasionally on TV weather casts goes something like this: “When the sun sets, the ground begins to emit infrared radiation...” That's just not how it works. The ground doesn't “start” emitting when the sun sets. Planck's Law tells us that the ground is always emitting infrared radiation, the amount and distribution versus wavelength of which depends upon its temperature, a fact that we'll explore later in this lesson.
- Wien's Law
So, Planck's Law tells us that all matter emits radiation at all wavelengths all the time, but there's a catch: Matter does not emit radiation at all wavelengths equally. This is where the next radiation law comes in. Wien's Law states that the wavelength of peak emission is inversely proportional to the temperature of the emitting object. Put another way, the hotter the object, the shorter the wavelength of maximum emission. You have probably observed this law in action all the time without even realizing it. Want to know what I mean? Check out this steel bar. Which end might you pick up? Certainly, not the right end! It looks hot, doesn't it? Why does it “look hot?”
 You Can Look (But You Better Not Touch)Credit: You Can Look by Caroline is licensed by CC 2.0 BY-NC-SA
You Can Look (But You Better Not Touch)Credit: You Can Look by Caroline is licensed by CC 2.0 BY-NC-SAWell, for starters, the peak emission for the steel bar (even the part that looks really hot) is in the infrared part of the spectrum. But, the right side of the bar is hotter than the left, and therefore the right side has a shorter wavelength of peak emission compared to the left side. You see this shift in the peak emission wavelength as a color change from red to orange to yellow as the metal's temperature increases. In fact, the right side is hot enough that its peak emission is pretty close to the visible part of the spectrum (which has shorter wavelengths than infrared); therefore, a significant amount of visible light is also being emitted from the steel.
Judging by the look of this photograph, the steel has a temperature of roughly 1500 Kelvin, resulting in a max emission wavelength of 2 microns (visible light has wavelengths of 0.4-0.7 microns). Here is a chart showing how I estimated the steel temperature.
 Steel color temperature chart
Steel color temperature chartTo the left of the visibly red metal, the bar is still likely several hundred degrees Celsius. However, in this section of the bar, the peak emission wavelength is far into the infrared portion of the spectrum, and no visible light emission is discernible with the human eye.
So, how do we apply Wien's Law to the emission sources that effect the atmosphere? Consider the chart below, showing the emission curves (called Planck functions) for both the sun and the Earth.
Note the idealized spectrum for the earth's emission of electromagnetic radiation (dark red line) compared to the sun's electromagnetic spectrum (orange line). The radiating temperature of the sun is 6000 degrees Celsius compared to the earth's measly 15 degrees Celsius. This means that, given its high radiating temperature, the sun's peak emission occurs in the visible light portion of the spectrum, near 0.5 microns (toward the short-wave end of the EM spectrum). That wavelength is also the reason why we see the sun as having a yellow hue. Meanwhile, the earth's peak emission is located in the infrared portion of the electromagnetic spectrum (having longer wavelengths, by comparison).
It is important to remember this relationship going forward. In understanding climate energy balance, it is very common for scientists to refer to radiation emanating from the sun as shortwave radiation, while radiation emanating from the Earth as longwave radiation. While we’ll stick to this convention in this class, you may also hear these referred to as solar (coming from the sun) and terrestrial (coming from the Earth) radiation, respectively. As seen below, these spectra have very little overlap, which is going to allow us to treat shortwave and longwave radiation distinctly from one another, which is going to make our energy budget calculations much easier!
- Stefan-Boltzmann Law
 The emission spectrum of the sun (orange curve) compared to the earth's emission (dark red curve). The x-axis shows wavelength in factors of 10 (called a “log scale”). The y-axis is the amount of energy per unit area per unit time per unit wavelength. I have kept the units arbitrary because they are quite messy. The important message is that the sun's emission spectrum peaks in the visible spectrum while the earth's emission spectrum peaks in the infrared (in accordance with Wien’s Law).Credit: David Babb @ Penn State is licensed under CC By-NC-4.0
The emission spectrum of the sun (orange curve) compared to the earth's emission (dark red curve). The x-axis shows wavelength in factors of 10 (called a “log scale”). The y-axis is the amount of energy per unit area per unit time per unit wavelength. I have kept the units arbitrary because they are quite messy. The important message is that the sun's emission spectrum peaks in the visible spectrum while the earth's emission spectrum peaks in the infrared (in accordance with Wien’s Law).Credit: David Babb @ Penn State is licensed under CC By-NC-4.0Look again at the graph of the sun's emission curve versus the earth's emission curve (above) and take note of the energy values on the left axis (for the sun) and right axis (for the earth). The first thing to notice is that the energy values are given in powers of 10 (that is, 10 6 is equal to 1,000,000). This means that if we compare the peak emissions from the Earth and sun, we see that the sun at its peak wavelength emits nearly 3,000,000 times more energy than the Earth at its peak. In fact, if we add up the total energy emitted by each body (by adding the energy contribution at each wavelength), the sun emits over 150,000 times more energy per unit area than the Earth!
I calculated the number above using the third radiation law that you need to know, the Stefan-Boltzmann Law. The Stefan-Boltzmann Law states that the total amount of energy per unit area emitted by an object is proportional to the 4th power of the temperature. You won't need to do any specific calculations with the Stefan-Boltzmann Law, but you should understand that as temperature increases, so does the total amount of energy per unit area emitted by an object (hotter objects emit more total energy per unit area than colder objects). This relationship is particularly useful when we want to understand how much energy the earth's surface emits in the form of infrared radiation. It will also come in handy when we study the interpretation of satellite observations of the earth, later on.
Think About It
Quiz Yourself...
What happens to that radiation, anyway?
What happens to that radiation, anyway?Prioritize...
After completing this section, you should be able to:
- describe and explain the effects of altitude on a location's average yearly temperatures.
- apply terms associated with vertical temperature variations, such as lapse rate, environmental lapse rate, and tropopause.
Read...
Now that we've covered the basic behavior of radiation and how it relates to temperature, we need to wrap-up our look at radiation by examining the possible fates of a “beam" of radiation as it passes through some sort of material.
When radiation first encounters some medium (whether it be a collection of gases, a liquid, or a solid), only three things can occur to that radiation. The electromagnetic energy can either be absorbed by the medium, scattered by the medium, or it can pass through the medium unaffected (a process called transmission). In many cases, all three processes can and do occur to some degree. Examine the figure below showing the three processes that can affect radiation passing through a medium.

Let's briefly discuss each of these potential outcomes:
- Transmission is essentially the process by which radiation passes through an object unaffected. An example of a medium with a high transmission value is window glass. Visible light passing through a thin sheet of glass does so basically undisturbed, which is why we can see objects clearly on the other side. We tend to call such mediums “transparent,” while mediums having low transmission values are called "opaque.” The transmission properties of a medium are highly dependent on wavelength, however. For example, an object that is transparent in the visible wavelengths might be opaque in some infrared wavelengths. I should also point out that 100% transmission is rare, except within the vacuum of space. Almost always, at least a little energy is lost to absorption and/or scattering as radiation moves through the medium. A glass of water from a clean lake may look clear, but it’s rare I can see more than a few feet down, no matter how pristine the water is.
- Absorption is the extinguishing of a portion of the radiation “beam.” When an object absorbs electromagnetic radiation, the radiation is taken up by the matter via increases in the rotational, vibrational, and/or electronic energies of its constituents. This increase in internal energy within the matter leads to a temperature increase of the matter. As with transmission, the amount of energy that an object absorbs depends on the wavelength of the radiation and the physical make-up of the object. For example, freshly fallen snow absorbs little direct sunlight, but snow readily absorbs infrared radiation.
- Scattering occurs when radiation interacts with matter in a way that changes its direction of “travel.” Scattering can occur in all directions, although some directions are preferred, depending on the size and composition of the particles involved in the scattering event. If the radiation encounters a scattering event and continues in a forward direction, the event is called “forward-scattering.” Likewise, objects can also back-scatter radiation, meaning that they redirect the radiation in all directions back toward the source. In some rare cases, the scattered radiation may retain the exact same direction that it initially had before the scattering event. When this occurs, the scattered light is sometimes counted in the “transmission” category (because it seemingly emerged unchanged from the medium).
Now, let's see these processes (particularly absorption and scattering) in action in the atmosphere. First, the atmosphere, like snow, is a highly discriminating absorber (it only absorbs certain wavelengths of the electromagnetic spectrum). The plot of absorption spectra by various gases (below) indicates how efficiently certain gases and the atmosphere, taken as a whole, absorb various wavelengths of electromagnetic radiation. To interpret the graph, note the “0 to 1” scale on the left of the plot, indicating zero percent absorption and 100 percent absorption, respectively. At any specific wavelength, the upward reach of the color shading indicates the percentage of absorption by a particular gas (or the atmosphere, taken as a whole).

For example, focus your attention on the row for oxygen and ozone, labeled “O2 and O3.” Note, to the left of the labels, that nearly 100 percent of the radiation incident on the O2 and O3 at wavelengths ranging from 0.1 microns to about 0.3 microns is absorbed. Recall that these wavelengths correspond to potentially dangerous ultraviolet radiation emitted by the sun. Ozone, a gas composed of three oxygen atoms (O3), absorbs much of the incoming ultraviolet radiation in the stratosphere, which is a layer that spans from 10 to 30 miles above the Earth's surface. Thank goodness for ozone in the stratosphere! Otherwise, cases of skin cancer and other afflictions associated with overexposure to the sun would likely be much more rampant in our society than they actually are.
Scattering, on the other hand, makes things look the way they do. You can't see objects if visible light isn't scattered to your eyes. But scattering doesn't have to be a one-time event. Often, radiation will enter an object and encounter many (hundreds/thousands) of scattering events before emerging. This is what happens to make clouds appear white on top and darker on the bottom (cue the obligatory storm photo). It's also what makes snow, salt, sugar, and milk white. Furthermore, multiple scattering increases the time that the radiation resides in the medium (as it bounces around, unable to escape). This longer residence time increases the chance that the radiation will also be absorbed by the medium. A great example is the blue hue that ice sometimes develops. Water (even in frozen form) tends to absorb wavelengths associated with red light at a faster rate than those associated with blue light, so over time with multiple scattering events, more blue light is scattered to our eyes (see below)!
Quiz Yourself...
Solar Radiation
Solar RadiationPrioritize...
By the end of this section, you should be able to:
- discuss how the sun’s energy is spread out over the spherical surface of the Earth,
- explain how the Earth's tilt on its axis of rotation impacts how directly this energy strikes the Earth throughout the year, and
- detail how seasons result from the changing angle at which the sun strikes the earth.
Read...
We now have a conceptual understanding of different types of radiation and what happens to radiation moving throughout a medium. But how does this pertain to the climate system?
OK, let’s start with where the energy actually comes from. By far, the most significant source of energy input into the Earth system is via solar radiation – i.e., the sun! It’s not the only energy source – others include geothermal energy, tidal energy, and energy from cosmic rays – but these are all much, much, smaller than the energy we get from our nearest star.
Recall that from above, the spherical sun emits a tremendous amount of energy into space. This energy is emitted isotropically (equally in all directions). As it travels out into space, it spreads out and the energy per unit area of space it passes through becomes less. Imagine blowing up a latex balloon – when you begin to inflate the balloon, the latex is still relatively “dense” and hard to see through. But as you keep blowing it up and spreading that material over larger and larger radii, the density becomes smaller, and the balloon thins. That “thinning” also happens with radiation density. It’s one of the reasons we are quite thankful we are not living on Mercury, where the energy density from the sun is much higher. The same goes for Pluto, too, which is very cold thanks to this process.
On Earth, at the top of the atmosphere (sometimes abbreviated TOA, you can think of this as the area in space just above all molecules in the Earth’s atmosphere), the solar flux density is – a value known as the solar constant. What does this tell us? Well, for our orbital radius, we get 1360 watts of energy input for every square meter (a two-dimensional square 1m on both sides) of that energy’s intersection with the Earth. A watt is just an energy flow per unit time – it is the same as Joules per second. So it’s kind of like measuring how much water is going through a hose and being sprayed on your lawn (well, if instead of gallons we talked about Joules!). We call this a constant because the sun is putting out almost the same amount of energy day-after-day, year-after-year. This varies a bit with sunspots every so often, but can be effectively thought of as something that will remain the same for our entire lifetime (more on its changes through the Earth’s history later!).
Because the Earth is a sphere, the actual average energy per unit area the Earth receives is a factor of four less. Why four you say? Well, imagine holding up a basketball and allowing your friend to shine a flashlight on it. For the person standing with the flashlight, the ball looks like a circle to them – the rays of light emanating from the flashlight get intercepted by the cross-sectional area of the ball. However, you (the one actually holding the ball) can see that the energy is actually not being spread across the “circle” that your friend sees, but rather the surface of the ball (for now, just the half facing the light, but if you spin the ball like the Earth rotates, eventually light is spread over the whole ball for each full rotation). Since the area of a circle is , but the surface area of a sphere is , the actual energy input the Earth receives per unit area is or . This is sometimes referred to as the solar “insolation” – think about it as Incoming SOLar radiATION.
To give you a visual, see the figure below. The yellow disk is what the sun sees Earth as -- a big circle. But we know that the Earth is a sphere, so the energy contained within this circle must be spread out over the surface area of the sphere. We just do our simple division above and get a factor of 4!

Latitudinal variations in solar insolation
We should note that this energy isn’t evenly spread over the surface, even with the planet rotating. The Earth is spherical (well, mostly spherical, although we’ll assume this is the case in this class) which means that as it orbits the sun, different latitudes (from the equator to the poles) receive sunlight at different angles. Why is this? Well, remember our basketball analogy from before – from the perspective of the sun, we see the Earth as just a circle that gets in the way of the rays we send out. Since the sun is quite far from the Earth, scientists make an assumption -- we assume the light is plane-parallel. We assume that every ray of light striking the Earth is moving parallel to all the other rays of light. This means that in areas where these solar rays are moving perpendicular to the Earth’s surface (toward the middle of our basketball or the Equator of the Earth), they are more concentrated in space. You can think of this as “more rays per square meter.” At high latitudes (towards the top and bottom of the basketball or our North and South Poles on Earth), these rays strike at an angle, which spreads them out across the surface (or disperses them). This means “fewer rays per square meter.” You can test this yourself at home right now if you want. Direct your attention to these side-by-side photos of a flashlight shining on a wall.

Please note that when the light from the flashlight strikes the surface at a rather direct angle, the light focuses on a rather small area. In other words, the light is intense. On the other hand, if the flashlight is tilted so that the light strikes the surface at a lower angle, the light spreads over a larger area, making the light less intense. Go find a dark room and a flashlight and try it out for yourself!
See the figure below for a visual interpretation.

What this means is over the course of a year, the Equator receives more radiation than higher latitudes. See the figure below, which shows the top of the atmosphere insolation. The average over this whole map is approximately (which we discussed above!), but there are higher values near the Equator where the sun’s radiation is most perpendicular and lower values towards the North and South Poles where the radiation strikes the planet at more oblique angles. Keep this geometry in mind – the fact that the Equator receives more energy from the sun than polar regions underpins why our atmosphere and ocean circulations behave the way they do.
Seasonal variations in solar insolation
What I showed you above is an annual average of radiation. Can you surmise why I did that? I deliberately left out one last important component of solar geometry –the tilt of the Earth's axis and Earth's orbit around the sun give rise to the seasons. If we didn’t have tilt, everything we talked about above would hold, but it’d be very close to the same temperature every day all year at a given spot on the planet. However, we know from personal experience in the United States that we aren’t receiving the same amount of energy all year long – we generally wear sunscreen more often in summer and usually must bundle up in winter. Why?
At the heart of Earth's seasonal variations is the tilt of its axis. The Earth’s axis can be shown by a line running from the North Pole to the South Pole, and the planet rotates around this axis like a toy top. But if an extraterrestrial alien were to stumble upon our planet in space, they’d realize that the poles are not perfectly up and down (or perpendicular) to its orbital plane, but rather the Earth is slightly tilted. In fact, it is inclined at an angle of approximately 23.5 degrees relative to its orbital plane. As Earth orbits the sun in an elliptical path, the orientation of its axis remains remarkably consistent – it’s always tilted at 23.5 degrees towards the North Star, no matter where it is in its orbit. This means that during different times of the year, specific parts of the Earth lean either toward or away from the sun.
When one hemisphere is tilted toward the sun, it receives sunlight that is more perpendicular to the surface, just like we discussed above. Like a nearly downward pointing flashlight shining directly on the surface from our dark room experiment, concentrated sunlight has heating power consistent with the elevated temperatures of summer. In the summer hemisphere, we also see that this is manifested in longer daylight hours and the sun can climb higher in the sky. You may not have considered this, but try to imagine where you see the sun in the sky during winter – it always feels like it’s not getting too far away from the horizon, even in the middle of the day! But you can certainly remember hot summer days when you craned your neck, stared straight up, and the sun was staring right back at you from directly overhead. This is the recipe for summer's warmth and extended days. In contrast, sunlight arrives at a more oblique angle when that hemisphere tilts away from the sun, leading to shorter days and colder temperatures. This means there are two days per year when the Earth is tilted either as far away or as close to the sun as possible. You may not know it, but you have heard of these days – they are the “solstices.” The periods when Earth's axis is neither tilted toward nor away from the sun are the transitional seasons of spring and autumn, when day and night lengths are relatively equal. There are two days every year when the Earth isn’t tilted away or towards the sun – these are the “equinoxes.”
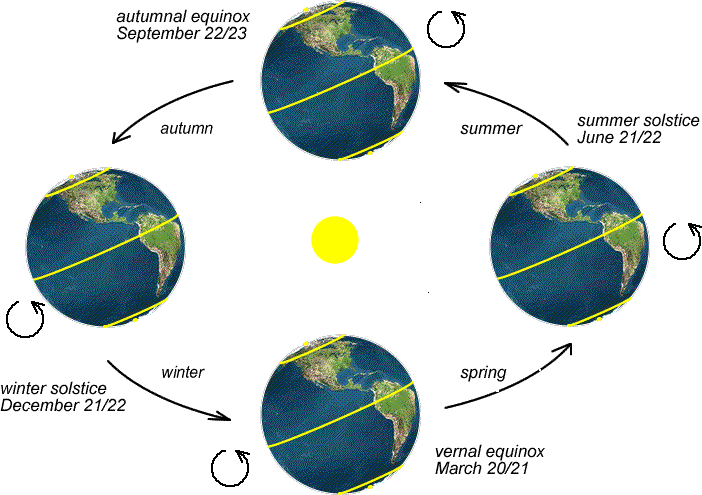
Think About It...
Do these maps make sense? Think about what the weather is like in December. Do you think your hometown gets more or less direct solar radiation during that month?
For now, we will focus on the annual averages to smooth out these seasonal variations. Still, we’ll return to the concepts of summer and winter and their importance for describing the temporal variations in climate later.
Fate of solar radiation striking the Earth
Since the sun is a constant emitter, the Earth is always receiving of energy at the top of the atmosphere. But why are we being so specific, saying the “top of the atmosphere” – that seems awfully suspicious. All that energy isn’t immediately transmitted straight to the surface – it can also be scattered or absorbed. So where does this energy go?
- About 30% is reflected back to space by air molecules, clouds, and the earth's surface. Note that I'm using the word “reflection” as a loose substitute for “back-scattering,” but there's a big difference between this loose use of “reflection” and the classic, pure interpretation of “reflection.” Pure reflection means that the angle at which radiation strikes an object must equal the angle at which the radiation is redirected from the object (think about how a billiard ball bounces off a bumper on a pool table).
- About 20% gets absorbed by clouds and atmospheric gases.
- Roughly 50% is transmitted and ultimately absorbed by the Earth's surface.

So, of the solar radiation reaching Earth's atmosphere, about 30% of it ends up back in space. It turns out we have a special name for the reflected portion -- albedo, which simply means striking some object that is ultimately not absorbed by it.
Albedo can vary between 0.0 and 1.0. An albedo close to 1.0 means almost 100% of the incoming solar energy is reflected away from the Earth system -- with respect to visible radiation, a mirror has an albedo close to 1.0. In contrast, an albedo close to 0.0 means almost all of it is being absorbed by the surface the radiation strikes. Think something matte black, like fresh asphalt.
On average, the Earth’s albedo is about 30 percent (0.3), but albedo over parts of the Earth varies greatly from location to location and time to time. Things that we consider “white” have high albedos. Two important surfaces that have high albedos are clouds and snow. Do you remember that sometimes your eyes hurt when walking outside on a sunny day after a fresh snowfall? Since snow has a high albedo, solar radiation is being reflected back towards your eyes, effectively giving you a “double whammy” of brightness – the sunlight coming down and the reflected radiation back up. Not a bad idea to keep those sunglasses handy in your glove box! On the other hand, surfaces like asphalt parking lots, forests, or ocean water tend to have low albedos because they don't reflect much of the solar radiation that strikes them. This is why it’s not pleasant to walk on asphalt on a sunny summer day.

Check out the map below, which shows the January surface albedo over the globe. For this figure, clouds have been removed – more on that in a second -- we are only considering the probability of sunlight being reflected at the surface on a sunny day. At high latitudes in the winter hemisphere (northern North America and Eurasia) albedo values are high, owing to snow and ice on the ground. The same holds for mountainous areas like parts of the Rocky Mountains, the Alps, and the Himalayas.

But wait a minute, why does Northern Africa have a similar albedo to the snowy high latitudes? Can you think of a major geographic feature in that area? Northern Africa is broadly covered by the Saharan Desert, which is generally devoid of large areas of water or vegetation. But sand is (perhaps somewhat surprisingly to you) a highly reflective surface for shortwave radiation from the sun. Next time you are at a large, sandy, beach, try and see whether you need your sunglasses more when compared to if you were walking in a grassy field or a forest nearby. You’ll find that your eyes behave very similarly to being over a snowy surface, and you’ll be squinting more when sitting in your lounge chair with your toes in the sand!
Now you may say, “Wait, if the sand in the Sahara reflects so much radiation, why is it so hot there?” That’s an excellent question, and we’ll answer that question in a few lectures – it’s the atmospheric circulation that plays a key role.

Quiz Yourself...
Clouds
CloudsPrioritize...
By the end of this page, you should be able to:
- Describe the basic process of cloud formation.
- List the three properties of a cloud important to determining its albedo.
Read...
In the last section, we said that white surfaces are key reflectors of solar radiation. In fact, we called out two particular surfaces, snow and clouds. While snow is generally seasonal and restricted to specific geographic regions, clouds are ubiquitous in our atmosphere. Check out the global satellite image below, it’s very rare to find spots on the Earth where there are no clouds nearby, and it’s even rarer for the sunny skies to remain day after day.
As you probably know from a previous science class, clouds are just condensed water. A simple explanation is that clouds are formed when warm, moist air rises and cools, causing water vapor to change from a gas to either a liquid or solid state.
As the moist, warm air containing water vapor rises, it cools down due to the decreasing air pressure at higher altitudes. As the air cools, it reaches a critical temperature called the dew point. The dew point is the temperature at which the air becomes saturated with moisture, meaning it can no longer hold all the water vapor in a gaseous state. When the air reaches its dew point, it becomes "saturated" with water vapor, and any additional moisture will condense into tiny water droplets or ice crystals, depending on the temperature. In most cases, air alone is not enough to initiate condensation. Tiny particles and aerosols in the atmosphere called condensation nuclei or cloud seeds play a crucial role. These particles can include dust, pollen, salt particles, and other atmospheric impurities. Water vapor condenses around these particles to form small water droplets. Once condensation begins around the condensation nuclei, it results in the formation of countless tiny water droplets or ice crystals (depending on the altitude where they are at and whether the temperature is broadly above or below freezing) suspended in the air. Collectively, these suspended water droplets or ice crystals form what we see as clouds.
Clouds can vary in appearance, shape, and altitude depending on factors such as temperature, humidity, and atmospheric conditions. They will be important in the context of the water cycle, which we’ll talk about soon, but it’s important to understand that this diversity in cloud properties can have a big role on their albedo and how much energy they scatter and absorb. Think about very thin wispy clouds you may see on a fair, otherwise sunny day. These are cirrus clouds which are made of ice crystals that are 100−200 µm in size and about 30,000 ice crystals per cubic meter (i.e., if you had a box that was one meter for each dimension, you’d have about 30,000 ice crystals in that box inside a cirrus cloud). This makes these types of clouds very transparent to sunlight – they don’t put up much of a fight when confronted with solar beams!

Conversely, we can have clouds that appear whiter in the sky. For example, cumulus clouds can also occur during general fair weather, but they are far more noticeable to you and me. That is because they are composed of water droplets that are roughly 10 microns in size. Wait, that is much smaller (by a factor of 10-20x!) than the cirrus clouds particles. How are cumulus clouds more “reflective” than cirrus clouds? The answer lies in the density of droplets that exist within that hypothetical one cubic meter box. The average cumulus cloud has somewhere around 65 million droplets per cubic meter! That high density of cloud particles makes it far more likely for sunlight to be reflected than if it was passing through a cirrus cloud. Now it makes sense when you climb through a cloud deck on your way out of an airport, and you suddenly can’t see anything but white outside your window.
So, the albedo of a cloud essentially depends on three things
- Mass of water in the cloud
- Droplet size
- Thickness of cloud
Numbers 1 and 2 are closely linked to the droplet density we discussed above. If we have a cloud with a lot of water and very small particles, it means a high number concentration, which reflects sunlight easily. On the other hand, a cloud with just a little bit of water tied up in big particles is relatively transparent to solar radiation. We didn’t talk about #3, but it’s pretty logical – if I take two of the same cloud and stack them right on top of one another, the thicker one will do a better job preventing radiation from being transmitted.
Now, we just talked about two types of clouds here, but there is a tremendous diversity of different cloud types we can’t cover in this class.
Quiz Yourself...
Budgets and Composition
Budgets and CompositionPrioritize...
After you've completed this section, you should be able to
- Describe how the Earth's energy budget is a balance between energy in and energy out
- List Earth's most abundant gases and be able to rank them.
Read...
Understanding the global climate system often involves thinking in terms of “budgets.” Traditionally, we've used the analogy of a checkbook to explain budgets, but in today's digital age, online bank accounts serve as a more relevant comparison. Imagine you have a certain amount of money saved up, let's say $1,000, in your bank account; this is your account balance. Now, there are two crucial aspects we need to consider: inflows and outflows. Inflows represent money being added to the bank account, while outflows represent money being withdrawn from it – a logical concept. To maintain a constant balance, your net inflow and net outflow should be equal, in other words, money in should equal money out.
The energy within the Earth’s climate system follows a similar principle. It's essential to remember that temperature is strongly connected to energy. Over the years, we've noticed that the average weather experienced in any given location on Earth remains relatively consistent in the long term – this is what we call climate. This consistency suggests that, to maintain global energy balance, the Earth must radiate longwave energy to space at the same rate that it receives shortwave energy from the sun!
Remember, the Stefan-Boltzmann Law tells us that *any* body with a temperature greater than zero must be emitting some energy. Given this, we can estimate Earth's global temperature using the information about solar radiation from the previous section and the Stefan-Boltzmann law within this straightforward in = out framework. Most of the energy entering the Earth's system comes from the sun. We've defined a solar constant () and explained that, in reality, this number gets divided by a factor of 4 when it's distributed across the entire surface of the Earth. Additionally, we've determined that Earth's albedo, representing the reflected energy, is approximately 30%. This means that only about 70% of the incoming radiation is absorbed by the planet, while the rest is reflected back into space. So, let's perform a simple calculation:
Therefore, when we average this value across the entire Earth, every square meter of the planet absorbs 239 Joules every second from the sun. That's a substantial amount of energy! A Tesla can go about 350 miles with about 8,000 W – think about this, if we managed to cover the playing field at Beaver Stadium (18,000 square meters) with perfect solar panels that took every bit of incoming solar energy and converted it to electricity, we could fully charge over 32,000 Teslas every single minute! This hopefully gives some indication why solar energy is considered such a promising renewable energy source.
This represents our energy input, but we also need to account for energy output. As mentioned earlier, the Stefan-Boltzmann Law relates the energy radiated from a black body in space to its temperature. To offset the incoming solar energy, Earth must radiate back into space. Without delving into too many details, we can use a simple equation to calculate this – don’t worry, I won’t make you solve it! The mathematical expression of Stefan-Boltzmann's law is as follows:
Here, E represents the emitted energy, T denotes the temperature, and σ is a constant (interestingly named the Stefan-Boltzmann constant). One thing it tells us is that as T goes up, E must also go up by some amount – we’ve already discussed that before. But if we rearrange the equation and solve for T, we can estimate the temperature Earth needs to have in order to radiate at and achieve our in = out balance. This is referred to as Earth's “emission temperature,” or in simpler terms, what an alien observer in outer space would measure with a thermometer pointed at Earth. If you're interested, feel free to use your calculators to verify, but the temperature comes out to be around 255 Kelvin. In more familiar units, this is equivalent to -18 °C or 0 °F. Quite chilly! We have satellites in space, and they corroborate this temperature as the overall emission temperature for Earth – fascinating, isn't it?
However, 0 °F is exceptionally cold! While certain regions on Earth can experience such cold temperatures occasionally, the average surface temperature of our planet is significantly warmer. Observations indicate that our global mean surface temperature is closer to 290 K (16.8 °C or 62 °F). This apparent disparity raises a crucial question: why is our planet's surface warmer than its emission temperature of longwave radiation to space? The answer lies in our atmosphere, which envelops our planet and makes the surface much more hospitable, given the input of solar energy. Continue reading to unravel this fascinating phenomenon.
Atmospheric composition
Before we explain why the Earth’s surface temperature is what it is, we need to briefly discuss the composition of the atmosphere. Obviously, the air we breathe is critical for our survival, but it's actually a mixture of many different types of molecules. Some of the gases are “permanent,” meaning that their concentrations are basically constant. Other gases are “variable,” meaning that their concentrations vary from time to time and place to place. I've summarized the gases that comprise our atmosphere and their concentrations in the table below:
| Permanent Gases | Variable Gases | ||
|---|---|---|---|
| Gas (Symbol) | Percent (by volume of dry air) | Gas (Symbol) | Percent (by volume) |
| Nitrogen (N2) | 78.08 | Water Vapor (H2O) | 0 to 4 |
| Oxygen (O2) | 20.95 | Carbon Dioxide (CO2) | about 0.041 |
| Argon (Ar) | 0.93 | Methane (CH4) | about 0.00018 |
| Neon (Ne) | 0.0018 | Nitrous Oxide (N2O) | about 0.00003 |
What are the big takeaways? First of all, even though we need to breathe oxygen to survive, oxygen is not the most abundant gas in the atmosphere. Nitrogen is, by far. There's nearly four times as much nitrogen as there is oxygen. However, nitrogen and oxygen, combined, account for roughly 99% of “dry air” in the atmosphere, so they're the “big two” in terms of total concentration. For every million molecules of air you breathe in, 990,000 of them are nitrogen and oxygen.
Of course, air isn't perfectly “dry.” Water vapor also exists in our atmosphere, but note that the concentration of water vapor is rather small and is variable (it varies from 0 to 4 percent). Furthermore, while you might hear a lot about carbon dioxide in the news because of its connection to climate change, it only accounts for about 0.041 percent of the atmosphere. Surprised? Give me a second, because we’ll talk about why it’s still important in a minute.
Quiz Yourself...
The "Greenhouse Effect” and our Final Energy Budget
The "Greenhouse Effect” and our Final Energy BudgetPrioritize...
Once you've completed this page, you should be able to
- discuss the so-called “greenhouse effect,” and the “greenhouse gases” that contribute to it, as well as its importance for life on Earth.
- describe the connection between the greenhouse effect and global warming, and make a distinction between the two.
Read...
So, we have this fundamental gap – out in space the Earth appears like it’s roughly 0F, but we know from living on the surface it’s generally warmer. Why? Well, recall that Planck’s Law told us that every piece of matter is emitting some form of energy. And the air molecules in our atmosphere are just that – it’s matter that can absorb and emit radiation itself. How does that help keep the surface temperature at a more comfortable level?
As you've learned, the earth's peak emission occurs at infrared wavelengths (from Wien's Law), so what happens to that radiation after it's emitted upward from the surface? Does it all go right to space? The answer is no; some is absorbed by air molecules, in particular, so-called “greenhouse gases,” such as water vapor, carbon dioxide, methane, and nitrous oxide. Note that I haven’t listed the oxygen and nitrogen discussed earlier – they are inefficient absorbers of longwave radiation and are quite happy to let that energy go back to space, not unlike how a clean window is quite happy to let shortwave radiation from the sun pass right through.
However, greenhouse gases are unique because they are a set of molecules that readily absorb longwave radiation that is emitted from the Earth. Of the greenhouse gases that exist within our atmosphere, water vapor is far and away the most abundant, followed by carbon dioxide (although recall that in the overall scheme of the atmosphere, these are trace gases – the percentages are very small). It turns out that some of the wavelengths that carbon dioxide and water vapor absorb readily (particularly those around 15 microns and a little larger) coincide with the wavelengths of Earth's peak emission.
Below is a chart of the absorption spectra of these “radiatively active gases” along with the atmosphere as a whole in the last row. Remember that shortwave radiation is anywhere between 0.1 and 1 µm. What this graph is showing us is that there is really not too much absorption in these wavelengths, so a lot of the curves are closer to zero than they are to one. The big exception is the third row, which is oxygen O2, and ozone O3. Ozone is the molecule in our atmosphere that most effectively absorbs shortwave radiation from the sun. This is actually a very important protectant for life on earth. Without ozone, a great deal of high intensity, ultraviolet (shortwave) radiation emitted by the sun would reach the surface, leading to far more frequent sunburns and skin cancer rates than seen today. We’ll talk about the ozone hole that formed a few decades ago and policies to protect our ozone layer later in the class.

Now focus on longer wavelengths, everything to the right of 1 µm. We see that all these gases (starting from the top, methane, nitrous oxide, ozone, carbon dioxide, and water vapor) all do some sort of absorbing in these wavelengths. That means, as the Earth emits these long wavelengths from the surface upwards, a select group of molecules in the atmosphere absorb some of this energy. But the molecules also must emit at these wavelengths – some of it is emitted upwards (towards space as before) but some of it is emitted downwards, back towards us at the surface. In fact, this downward emission is typically termed “down welling longwave” since it’s the terrestrial radiation that is emitted from the atmosphere back towards the surface of the Earth.
The fact that greenhouse gases absorb and emit infrared radiation so readily works out very well for humans. The very existence of these gases in our atmosphere is the key difference in why the Earth’s surface is actually that 62F number we discussed earlier versus the 0F we estimated it would be if there were no gases at all! So aside from all the oxygen we breathe, we can also thank our atmospheric blanket for making sure we don’t live in a total icebox!
The contributions of down welling IR from greenhouse gases to warming the planet is called the greenhouse effect. It is critically important to note that these gases are part of the natural Earth system – we’ll talk about human-induced global warming later, but it’s the increase in these greenhouse gases that is concerning, not their existence themselves!
Now we finally have all the pieces to explain the large-scale energy budget of the earth. When we average over the entire surface of the Earth, the sun provides approximately 340 W/m2 of incoming energy. Approximately 1/3 of that is reflected by either clouds or the surface back to space, thanks to our planetary albedo. Of the 2/3 left over (), is absorbed by the atmosphere on the way down (primarily the ozone we discussed earlier), and is absorbed by the surface. Most of the energy associated with the solar radiation absorbed in the surface is subsequently re-emitted by the surface as long wave radiation upwards into the atmosphere, although some of it also leaves the surface via other heat fluxes. Of the energy emitted by the surface, only approximately 5% of it makes it directly to space, with the other 95% being absorbed somewhere in the atmosphere, thereby changing atmospheric temperatures throughout. At the same time, all of the atmospheric volumes absorbing radiation from the surface are also emitting radiation according to their own temperatures. Eventually, the atmosphere exhausts to space via emission from gas molecules. When combined with the making it straight through, through to space from the surface, a total of 239 W / m2 is emitted to space as longwave radiation. This is quite close to the that comes into the Earth system from the sun and is absorbed by the Earth system!

Checkout this video (4:07 minutes) for a walkthrough of the energy flow diagram.
Walkthrough of the Energy Flow Diagram
Proffessor Colin Zarzycki: Let’s take a look at this energy budget in a little more detail. When we account for the curvature of the Earth, we get 340 watts per meter squared that is coming down from the sun. We know that before any of that energy is actually absorbed by material in the Earth system, a large fraction of it is reflected back to space. In fact, 100 watts per meter squared, and note that 100 watts per meter squared is approximately 29% of 340. This is our planetary albedo.
Now, of that radiation that is reflected back to space, 75 watts per meter squared are reflected by clouds, and 25 watts per meter squared are reflected by the Earth’s surface. Now, if you do 340 minus 100, that gives us 240 watts per meter squared. That’s the amount of energy from the sun that must be absorbed by components in the Earth system. 80 watts per meter squared are absorbed directly in the atmosphere, and 160 watts per meter squared are absorbed by the surface.
So now we know that we have 240 watts per meter squared of energy that is absorbed by the Earth system. Where does that energy go? Well, we can look at the surface energy budget. So remember, in accordance with Stefan-Boltzmann law, anything that has a nonzero temperature—thankfully, everything on Earth—is emitting some form of longwave radiation. So the Earth is constantly emitting radiation in the form of things like thermals. This is kind of sensible heat, heat that is directly being transmitted to the atmosphere. We also have evapotranspiration, so heat that is associated with things like the phase changes of water.
We’ll talk a little bit more about this, but the vast majority of energy that is emitted from the surface is longwave infrared energy that is emitted again in accordance with Stefan-Boltzmann law. Of that 396 watts per meter squared, 20 of it goes to space, but 376 watts per meter squared are actually absorbed in the atmosphere. And that atmosphere is allowed to then emit radiation both upward and downward. In fact, this back radiation back to the surface that the atmosphere is constantly emitting, again because it is a nonzero temperature, is what leads to the greenhouse effect that we discussed. This keeps our planet at a warmer temperature than we would expect just based on the total amount of solar energy in and the amount of energy out.
Now, the atmosphere itself at the end of the day is going to emit 219 watts per meter squared. And so when you combine 219 plus the 20 watts per meter squared that are emitted directly from the surface to space, you get a total outgoing longwave radiation at the top of the atmosphere of 239 watts per meter squared. This is almost exactly the same as the 240 watts per meter squared that we’re bringing in. So why is it off by this small amount? Well, there’s this one little arrow that we’re going to talk about later in the semester with regard to additional energy that is trapped or put into the system. But otherwise, you’ve now been able to account for where energy is moving within the Earth system and how to balance the amount of energy coming in from the sun and the amount of energy that the Earth is giving off in terms of its longwave radiation.
Quiz Yourself…
Latitudinal Distribution of Solar, IR, and Net IR
Latitudinal Distribution of Solar, IR, and Net IRPrioritize…
After reading this section, you should be able to:
- Explain how the distribution of solar (shortwave) radiation varies globally
- Understand the distribution of longwave radiation emitted by Earth's surface and atmosphere
- Describe the concept of net radiation
Read...
One last point we should make about radiation – how it is distributed globally is going to be quite important in the rest of this course. That is, we’ve been treating the Earth as a giant box for the most part – energy comes in, energy goes out. And that’s not a bad way to look at things when we talk about the Earth as a whole. But when we want to learn more about why the Sahara is so hot, the Arctic is so cold, why Cancun is such a popular spring break destination and Nome, Alaska isn’t, we need to start thinking about how radiation varies across the planet.
Shortwave Radiation Distribution:
We’ve already talked about this previously, so I won’t belabor it, but recall that we have more solar insolation at lower latitudes than higher ones. This is a fundamental result of the planetary tilt and the fact that the Earth is a sphere. Solar radiation is most intense near the equator, where sunlight strikes the Earth nearly perpendicular to the surface. As we move towards higher latitudes, the same amount of solar energy is spread over a larger area, resulting in less intense solar radiation. Consequently, regions near the poles receive considerably less solar energy than equatorial regions.
Longwave Radiation Distribution:
On the other end of the spectrum (no pun intended!), we have longwave radiation, which is emitted by the Earth's surface and its atmosphere. Recall that the Stefan-Boltzmann Law tells us that this emission is a direct consequence of Earth system temperatures – the hotter something is, the more longwave radiation it emits, the cooler something is the less longwave radiation it emits. Just as with solar radiation, the distribution of emitted longwave radiation varies with latitude. Warmer equatorial regions emit more longwave radiation, while cooler polar regions emit less.
Net Infrared Radiation Distribution:
So it seems like all the energy is going into and out of the tropics! That is true; low latitudes are critical for acting as the bank tellers for most of the Earth’s incoming and outgoing radiation. However, another way to look at this is through net radiation. Climate scientists love to create budgets, and one thing we can look at is the amount of energy in versus the energy out at each latitude. We know that the answer must be zero for the Earth system as a whole, but that doesn’t mean it has to be zero everywhere on the planet.

Quiz Yourself...
Summary
SummaryRead…
- Energy is fundamental to understanding the climate system. It governs processes like movement, heat transfer, and changes in the Earth's atmosphere.
- Key principles: energy can be stored, transferred between objects, and transformed between different types like radiant, kinetic, gravitational potential, and internal energy.
- The First Law of Thermodynamics teaches us that energy is conserved — it can move around and change forms but cannot be created or destroyed.
- The Second Law of Thermodynamics explains how energy flows from hot to cold, increasing disorder (entropy) in the process.
- Radiant energy, especially from the sun, plays a central role in driving the Earth’s climate system, impacting how energy is stored, moved, and transformed on Earth.
- The Earth receives energy from the sun and radiates it back to space. This energy balance determines the planet's overall temperature and climate patterns.
- Greenhouse gases in the atmosphere both absorb and emit longwave radiation. The downward longwave radiation emitted by them helps to maintain a surface temperature much greater than the emission temperature of Earth, a phenomenon known as the greenhouse effect.
We also discussed how water vapor is the Earth's most abundant greenhouse gas. We also know from personal experience that water is critical to the climate. So, what role does water play, and how does it move around the climate system? Why are some regions dry and others wet? Next lesson, we'll cover that!


